Innovative cartilage implant for the treatment of tissue defects
Meidrix is specialized in the regenerative treatment of cartilage defects in joints such as knee, hip, shoulder, toe or ankle. The medical biotechnology company has developed the patented and CE-certified ChondroFiller, which is manufactured under GMP conditions in the variants ChondroFiller gel and ChondroFiller liquid.
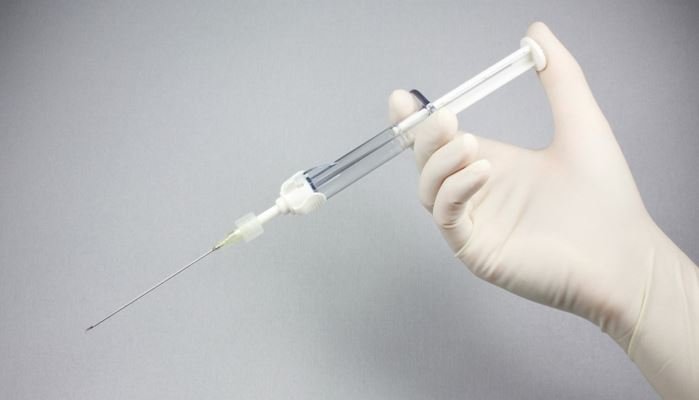
ChondroFiller liquid is a highly innovative collagen-based cartilage implant for the arthroscopic treatment of articular cartilage defects by means of using a unique, patented two-chamber syringe. The implant is liquidly applied and forms a solid collagen scaffold directly in the defect. The implant, which is indicated for clearly limited cartilage damage in different joints, enables the regenerative defect filling and the protection of the defect zone at the same time.
The three-dimensional matrix of the implant is based on animal collagen type I. It is a protein that is found in connective tissue, skin, tendons and bones and limits the elasticity of the tissues due to its tensile strength. It is one of the most important matrix proteins in the body and is characterized by a very good biocompatibility.
In the patient's body, the ChondroFiller implant provides a basic scaffold that allows colonization of the defect with the body’s own cells. Locally, the cells can now proliferate and start their cell-characteristic synthesis performance and form collagen type II, which is the major component of cartilage. MRI scans of first clinical trials show that cartilage defects treated with ChondroFiller are hardly to distinguish after six months from healthy cartilage.
Ahead of conventional forms of therapy
The cell-free, biological implants represent a new generation in the field of biomedicine. In contrast to conventional cartilage treatments, such as bone-cartilage transplantation or cartilage cell transplantation, there is no need for expensive cell collection in patients and long waiting times for cell cultivation. For complete defect filling, only one surgical procedure is required. Thus, this therapy is significantly cheaper and time-saving from which, in the end, patient, doctor and health insurance benefit.
Further information can be found at www.meidrix.de.